Terazosin Hydrochloride Dihydrate
Price 100 INR/ Kilograms
Terazosin Hydrochloride Dihydrate Specification
- Poisonous
- No (when used as directed in medical use)
- Molecular Weight
- 459.95 g/mol (base molecule), dihydrate salt form may differ
- Smell
- Odorless
- Shelf Life
- 2-3 years if properly stored
- Boiling point
- Not applicable (decomposes)
- EINECS No
- 264-079-2
- Color
- White
- Solubility
- Soluble in water, practically insoluble in ether
- HS Code
- 29420090
- Heavy Metal (%)
- 0.001%
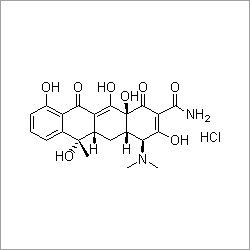
- Structural Formula
- See image (https://cpimg.tistatic.com/4501267/s/4/Terazosin-Hydrochloride-Dihydrate.gif)
- Loss on Drying
- 1.0%
- Molecular Formula
- C19H25N5O4S2HCl2H2O
- Storage
- Store between 2C to 8C in a tightly closed container, protected from light and moisture.
- Melting Point
- 273-275C (dec.)
- Medicine Name
- Terazosin Hydrochloride Dihydrate
- Chemical Name
- Terazosin Hydrochloride Dihydrate
- CAS No
- 63590-64-7
- Type
- Other
- Grade
- Other
- Usage
- Terazosin Hydrochloride Dihydrate is used primarily as an antihypertensive agent and to treat benign prostatic hyperplasia (BPH).
- Purity(%)
- >=98.0%
- Appearance
- White or almost white powder
- Physical Form
- Powder
- Standard
- Complies with in-house specifications or pharmacopeial standards upon request
- Sensitivity
- Sensitive to moisture and light
- Formulation
- API for solid oral dosage forms
- Identification Tests
- By IR, HPLC, and chemical methods
- Residual Solvents
- Meets ICH guidelines
- Packaging
- Sealed in HDPE bottles or triple-layer aluminum bags
- Related Substances
- Complies with pharmacopeial requirements
- Assay
- >=98.0% (on anhydrous basis)
- Impurities
- Meets applicable impurity profile limits
Terazosin Hydrochloride Dihydrate Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Terazosin Hydrochloride Dihydrate
Application and Usage Guidance
Terazosin Hydrochloride Dihydrate is expertly formulated for use in medicinal plant environments as an Active Pharmaceutical Ingredient (API) for solid oral dosage forms. It is suitable for treating hypertension and benign prostatic hyperplasia (BPH) only through authorized medical channels. Its specific chemical profile and moisture sensitivity make it most effective when handled in properly equipped pharmaceutical production facilities. Manufacturers, distributors, exporters, and suppliers rely on its flawless performance and consistent results.
Order, Payment, and Packaging Information
Dispatched swiftly upon order completion, Terazosin Hydrochloride Dihydrate is delivered securely in HDPE bottles or triple-layer aluminum bags, ensuring optimal protection. Payment terms are flexible and can be tailored based on contract; samples are available to facilitate your quality checks prior to bulk purchases. Every order undergoes rigorous documentation, packaging, and shipping procedures to maintain its first-rate integrity throughout the distribution process.
FAQs of Terazosin Hydrochloride Dihydrate:
Q: How should Terazosin Hydrochloride Dihydrate be stored to maintain its quality?
A: To preserve its purity, store Terazosin Hydrochloride Dihydrate between 2C to 8C in tightly closed containers, protected from light and moisture.Q: What is the primary medical usage of Terazosin Hydrochloride Dihydrate?
A: Terazosin Hydrochloride Dihydrate is primarily utilized as an antihypertensive agent and for the treatment of benign prostatic hyperplasia (BPH).Q: When can I expect my order of Terazosin Hydrochloride Dihydrate to be dispatched?
A: Orders are dispatched promptly after completion and approval, with delivery timelines depending on shipping location and contract terms.Q: Where can I request a Terazosin Hydrochloride Dihydrate sample prior to bulk purchase?
A: You can request a sample directly through the suppliers ordering process to verify quality and suitability before making a bulk commitment.Q: What identification tests are performed on Terazosin Hydrochloride Dihydrate?
A: Identification is confirmed via IR, HPLC, and chemical testing methods to guarantee compliance with pharmacopeial standards.Q: How does Terazosin Hydrochloride Dihydrate ensure safety regarding residual solvents and heavy metals?
A: It meets stringent ICH guidelines for residual solvents and maintains heavy metal content at 0.001%, enhancing safety for medicinal use.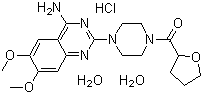

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
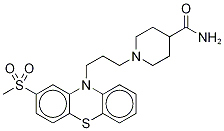
Metopimazine API
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Heavy Metal (%) : 10 ppm max
HS Code : 29349990
Solubility : Soluble in water and ethanol
Color : White to offwhite, Other
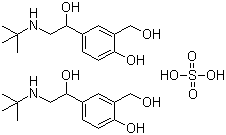
Albuterol Sulfate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Heavy Metal (%) : 0.001%
HS Code : 29372900
Solubility : Freely soluble in water, slightly soluble in ethanol
Color : White
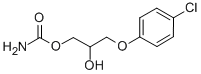
Chlorphenesin Carbamate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Heavy Metal (%) : 0.001%
HS Code : 29241900
Solubility : Soluble in ethanol and chloroform, slightly soluble in water
Color : Other, White to offwhite
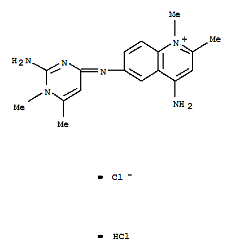
Quinapyramine Sulphate
Price 100 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Heavy Metal (%) : <0.001%
HS Code : 29349900
Solubility : Soluble in water
Color : Other, White to offwhite
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry