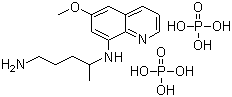
Ibutilide fumarate
Price 100 INR/ Kilograms
Ibutilide fumarate Specification
- HS Code
- 29349900
- Loss on Drying
- Not more than 0.5%
- Boiling point
- Not applicable / decomposes
- Shelf Life
- 2 years if properly stored
- Melting Point
- 131-135C
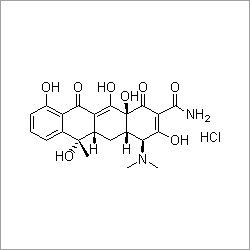
- Structural Formula
- See image or chemical structure
- Heavy Metal (%)
- 20 ppm
- Molecular Formula
- C23H36N2O5S C4H4O4
- Solubility
- Freely soluble in water, sparingly soluble in methanol
- Smell
- Odorless
- Storage
- Store at 2-8C, protected from light and moisture
- Molecular Weight
- 566.7 g/mol (fumarate salt)
- Color
- White to off-white
- Poisonous
- Not classified as poisonous, but intended for pharmaceutical use only
- Medicine Name
- Ibutilide Fumarate
- Chemical Name
- ()-N-{4-[4-(Ethylheptanoylamino)-1,2,3,4-tetrahydrophenyl]butyl}methanesulfonamide fumarate
- CAS No
- 138402-11-6
- Type
- Other
- Grade
- Other
- Usage
- Pharmaceutical intermediate; antiarrhythmic agent
- Purity(%)
- >=98%
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Identification
- By IR, HPLC retention time
- Chirality
- Racemic mixture ()
- MSDS
- Available on request
- Packing
- In double polyethylene bags, placed in fiber drums
- Certificate of Analysis
- Provided with each batch
- End Use
- For formulation of intravenous infusion for acute arrhythmias
- Impurity Level
- NMT 0.1% for any individual impurity
- Therapeutic Category
- Class III antiarrhythmic
- Manufacturing Standard
- cGMP compliant
- Residual Solvents
- Complies with ICH guidelines
Ibutilide fumarate Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Ibutilide fumarate
Competitive Edge & Usage Benefits
Ibutilide Fumarate offers remarkable reliability for pharmaceutical manufacturers, featuring an impurity level NMT 0.1% and robust cGMP compliance. Its primary use is in formulating intravenous infusions for acute arrhythmias, serving hospitals, clinics, and research facilities. The compounds advanced certification and analysis ensure consistent quality. Its elite properties make it preferred by top-tier distributors, exporters, and importers seeking assured efficacy and safety in their supply chain.
Packaging, Sample Policy & Price Advantages
Ibutilide Fumarate is securely packed in double polyethylene bags within fiber drums, ensuring safe exchange and maintaining product integrity during transport and storage. Samples are available on request, following a straightforward quotation and sample policy, designed to provide the lowest price without compromise on quality. Potential clients can easily confirm product performance and compliance before purchase, offering confidence with every transaction.
FAQs of Ibutilide fumarate:
Q: How is Ibutilide Fumarate identified and tested for quality?
A: Ibutilide Fumarate is identified using IR and HPLC retention time methods. Each batch is accompanied by a Certificate of Analysis and meets cGMP standards, with impurity levels not more than 0.1%, offering top-tier quality assurance.Q: What are the recommended storage conditions for Ibutilide Fumarate?
A: Ibutilide Fumarate should be stored at 2-8C, protected from light and moisture. These conditions optimize shelf life and maintain product stability for up to two years.Q: When can a sample of Ibutilide Fumarate be requested and what is the policy?
A: Samples of Ibutilide Fumarate can be requested anytime in the quotation exchange process. The sample policy is client-friendly, allowing product evaluation before bulk purchase at the lowest price.Q: Where is Ibutilide Fumarate primarily applied?
A: Ibutilide Fumarate is primarily used in hospitals, clinics, and pharmaceutical R&D for the intravenous formulation of antiarrhythmic agents treating acute arrhythmias.Q: What are the main benefits of choosing Ibutilide Fumarate for pharmaceutical manufacturing?
A: Choosing Ibutilide Fumarate guarantees a high-purity, elite-grade intermediate with strict compliance to ICH guidelines and cGMP manufacturing, ensuring safety and effectiveness in finished pharmaceutical products.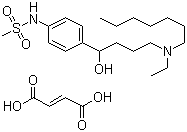

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Gliclazide
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Gliclazide
Grade : Other
Purity(%) : >=99%
Storage : Other, Store in a cool, dry place, protected from light and moisture
Primaquine Phosphate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : N4(6Methoxyquinolin8yl)1,4pentanediamine phosphate
Grade : Other
Purity(%) : 98%
Storage : Other, Store in a cool, dry place, tightly closed container, protected from light
Robenacoxib .
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Robenacoxib
Grade : Other
Purity(%) : 98%
Storage : Other, Store at 28C, keep container tightly closed
Cetrorelix Acetate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Cetrorelix Acetate
Grade : Other
Purity(%) : >=98%
Storage : Other, Store at 20C, protect from light and moisture
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry