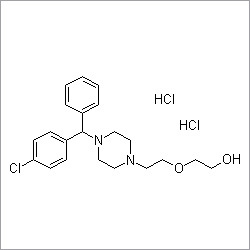
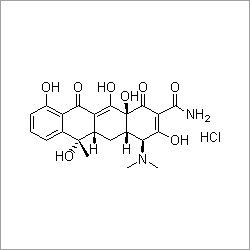
Pitavastatin Calcium
Price 100 INR/ Kilograms
Pitavastatin Calcium Specification
- Poisonous
- NO
- Structural Formula
- See image or as per chemical structure
- Storage
- Store in a cool, dry and well-ventilated place, protected from light
- Boiling point
- Not applicable (decomposes before boiling)
- Solubility
- Slightly soluble in water; soluble in methanol and DMSO
- HS Code
- 29420090
- Smell
- Odorless
- Melting Point
- 181-185C
- Heavy Metal (%)
- 20 ppm
- Shelf Life
- 2 years
- Molecular Formula
- C50H46CaF2N2O8
- Loss on Drying
- 0.5%
- Color
- White
- Molecular Weight
- 881.02 g/mol
- Medicine Name
- Pitavastatin Calcium
- Chemical Name
- (3R,5S,6E)-7-[2-Cyclopropyl-4-(4-fluorophenyl)quinolin-3-yl]-3,5-dihydroxyhept-6-enoic acid calcium salt
- CAS No
- 147520-94-5
- Type
- Other
- Grade
- Other
- Usage
- Used as a lipid-lowering agent for treatment of hypercholesterolemia and dyslipidemia
- Purity(%)
- >99%
- Appearance
- White or almost white powder
- Physical Form
- Solid
- Microbial Limit
- Total bacterial count <1000 CFU/g, absence of pathogens
- Stability
- Stable under recommended storage conditions
- Packing
- Packed in double polyethylene bags placed in aluminum foil bags and further in fiber drums
- Identification Methods
- IR, HPLC, Mass Spectrometry
- Related Substances
- 0.2%
- Pharmacopoeial Status
- Available as per in-house specifications or on request USP/EP/JP
- Assay
- >99.0% (by HPLC)
- Impurity Content
- 0.2%
- Residual Solvents
- Complies with ICH guidelines
Pitavastatin Calcium Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Pitavastatin Calcium
Other Applications and Material Features of Pitavastatin Calcium
Beyond its primary use in cholesterol management, Pitavastatin Calcium is leveraged in advanced pharmaceutical formulations and research as an active compound. Its solid form, exquisite purity, and compliance with safety benchmarks make it suitable for a variety of dosage preparations. The materials perfect stability, odorless nature, and double-layered packaging ensure optimal handling and extended shelf life, establishing it as a top choice for distributors, manufacturers, and research laboratories alike.
Sample Availability, Payment Terms, and Certifications for Pitavastatin Calcium
A sample of Pitavastatin Calcium can be quickly handed over for quality evaluation before any bulk order is made. Products are dispatched only after documentation and certification requirements are fulfilled, ensuring regulatory compliance. Payment terms are flexible, tailored to client-specific contracts or purchase orders. Full quality certifications are presented at handover, confirming adherence to pharmacopoeial standards and industry benchmarks. Timely and secure delivery is guaranteed for all transactions.
FAQs of Pitavastatin Calcium:
Q: How is Pitavastatin Calcium identified and tested for quality assurance?
A: Pitavastatin Calcium is identified and confirmed using IR, HPLC, and mass spectrometry. Each batch undergoes rigorous assay testing and impurity analysis to verify a purity of over 99% and ensure related substances do not exceed 0.2%.Q: What are the recommended storage conditions for Pitavastatin Calcium?
A: Pitavastatin Calcium should be stored in a cool, dry, and well-ventilated environment, protected from light to maintain its stability and two-year shelf life.Q: When can a sample of Pitavastatin Calcium be requested and handed over?
A: A sample of Pitavastatin Calcium can be requested at any time during the purchase inquiry process. It will be handed over following necessary documentation and approval procedures.Q: Where is Pitavastatin Calcium typically used?
A: Pitavastatin Calcium is commonly used in pharmaceutical manufacturing for lipid-lowering medicines and in research laboratories for studying cholesterol metabolism and related formulations.Q: What benefits does Pitavastatin Calcium offer regarding safety and compliance?
A: Pitavastatin Calcium meets strict safety and compliance standards, including low microbial count, absence of pathogens, and conformity with ICH guidelines on residual solvents and heavy metals, ensuring it is safe and reliable for pharmaceutical use.Q: Is Pitavastatin Calcium poisonous or hazardous?
A: No, Pitavastatin Calcium is not classified as poisonous. It is odorless and meets all prescribed safety and impurity limits, making it suitable for regulated pharmaceutical applications.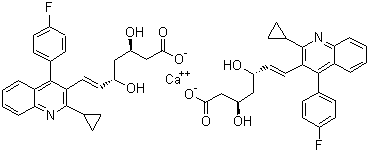

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
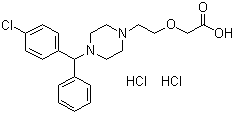
Cetirizine dihydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Poisonous : Other, Nonpoisonous at therapeutic dose
Structural Formula : C21H27Cl3N2O3
Storage : Other, Store in a cool, dry place, tightly closed and protected from light
Solubility : Freely soluble in water, sparingly soluble in methanol, practically insoluble in acetone
Triamterene
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Poisonous : Other, Nonpoisonous at therapeutic doses
Structural Formula : See image or: C12H11N7
Storage : Other, Store in a cool, dry, wellventilated area. Keep container tightly closed.
Solubility : Insoluble in water; slightly soluble in alcohol, chloroform.
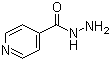
Isoniazid .
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Poisonous : Other, Yes, if taken in excessive quantities
Structural Formula : C6H7N3O
Storage : Other, Store in a dry, cool place, protected from light
Solubility : Freely soluble in water; slightly soluble in ethanol
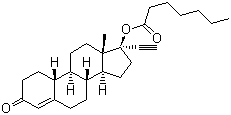
Norethisterone Enanthate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Poisonous : Other, No (when used at recommended doses)
Structural Formula : See chemical structure in reference image or catalog
Storage : Other, Store at 28C, protect from light and moisture
Solubility : Practically insoluble in water, soluble in methanol and ethanol
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry