Cefoperazone sodium
Price 100 INR/ Kilograms
Cefoperazone sodium Specification
- Loss on Drying
- NMT 3.0%
- Molecular Weight
- 667.65 g/mol
- Storage
- Store in a cool, dry place, protected from light
- EINECS No
- 263-894-7
- Molecular Formula
- C25H26N9NaO8S2
- Solubility
- Soluble in water
- Ph Level
- 4.5-6.5 (5% solution)
- Melting Point
- 214-216C (decomposes)
- Heavy Metal (%)
- NMT 0.001%
- Color
- White to off-white
- Boiling point
- Not applicable (decomposes before boiling)
- Particle Size
- As per manufacturer specification (typically fine powder)
- HS Code
- 29419060
- Smell
- Odorless
- Poisonous
- No (when used as directed)
- Shelf Life
- 2 years
- Taste
- Bitter
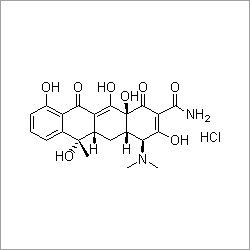
- Structural Formula
- C25H26N9NaO8S2
- Medicine Name
- Cefoperazone Sodium
- Chemical Name
- Cefoperazone Sodium
- CAS No
- 62893-19-0
- Type
- Other
- Grade
- Other
- Usage
- Used in the treatment of bacterial infections, particularly those caused by Gram-negative organisms
- Purity(%)
- >98%
- Appearance
- White to off-white powder
- Physical Form
- Powder
- Assay (on dried basis)
- >98.0%
- Water Content
- NMT 5.0% by Karl Fischer
- Reconstitution
- Reconstitute with sterile water for injection
- Category
- Third-generation cephalosporin antibiotic
- Identification
- Complies as per standard
- Related Substances
- NMT 1.0%
- pH of Injection Solution
- 4.5 to 6.5
- Packaging
- Available in vials
- Endotoxin Level
- NMT 0.2 EU/mg
- Microbial Limits
- Complies with standard pharmacopeia requirement
- Stability
- Stable under recommended storage conditions
- Residual Solvents
- Complies with USP/NF
Cefoperazone sodium Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Cefoperazone sodium
Enhanced Features & Versatile Applications
Cefoperazone sodium demonstrates remarkable compatibility with plant-based pharmaceutical operations, meeting stringent standards for heavy metals, water content, and related substances. Its main application is in treating severe bacterial infections, particularly those involving Gram-negative bacteria. This fine, white to off-white powder is effortlessly administered following reconstitution with sterile water. Its versatility extends to various medical facilities and pharmaceutical manufacturers, making it a preferred choice for both acute care settings and ongoing infection management.
Export Markets, Domestic Reach & Sample Quotation
Cefoperazone sodium enjoys substantial demand across global export markets and remains highly sought after in the domestic mar. Distributors, exporters, and suppliers offer prompt quotations tailored to the specific outlay expectations of clients worldwide. Samples are available upon request, facilitating product evaluation and compliance checks. Whether you operate in international pharmaceutical trade or local distribution networks, this product stands as a reliable, effective option to support your clinical and commercial objectives.
FAQ's of Cefoperazone sodium:
Q: How should Cefoperazone sodium be reconstituted for injection?
A: Cefoperazone sodium should be reconstituted using sterile water for injection, following manufacturer guidelines to ensure proper solubility and efficacy prior to administration.Q: What makes Cefoperazone sodium a preferred first-string antibiotic?
A: Its exceptional purity (>98%), stability under recommended storage, compliance with USP/NF standards, and proven effectiveness against Gram-negative organisms make Cefoperazone sodium a first-string choice in critical care hospitals and clinics.Q: When is Cefoperazone sodium most commonly used?
A: This antibiotic is primarily used in the treatment of severe bacterial infections, particularly those resistant to other treatments or caused by Gram-negative bacteria.Q: Where is Cefoperazone sodium typically supplied and used?
A: It is distributed to hospitals, clinics, and pharmaceutical manufacturers both domestically and internationally, and utilized across acute care and specialty medical environments.Q: What are the benefits of purchasing Cefoperazone sodium from your distribution network?
A: Buyers benefit from competitive quotas, reduced prices, strict compliance with international standards, available sampling, and consistent supply, making procurement efficient and dependable.Q: What is the process for obtaining a quotation or sample of Cefoperazone sodium?
A: Interested buyers can request a quotation or order a sample directly from accredited distributors or manufacturers, ensuring all regulatory and compliance documentation is included.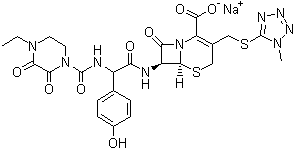

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
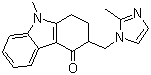
Ondansetron Base
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Grade : Other
Chemical Name : Ondansetron
Structural Formula : See Image
Shelf Life : 3 Years
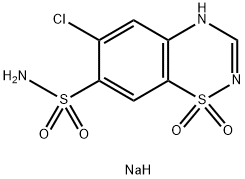
Chlorothiazide Sodium
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Grade : Other, USP Grade
Chemical Name : Sodium 6chloro2H124benzothiadiazine7sulfonamide 11dioxide
Structural Formula : C6H5ClN3O4S2Na
Shelf Life : 2 years from the date of manufacture when stored properly
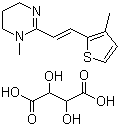
Morantel Tartrate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Grade : Other
Chemical Name : ()2,3,5,6Tetrahydro2,6dimethyl1H,1,3,5thiadiazine4one, Hydrogen Tartrate (1:1)
Structural Formula : Available on request
Shelf Life : 2 years
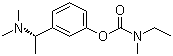
Rivastigmine
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Grade : Other
Chemical Name : Rivastigmine
Structural Formula : (refer to image or file for structure)
Shelf Life : 2 years
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry