Levetiracetam Api
Price 100 INR/ Kilograms
Levetiracetam Api Specification
- Molecular Formula
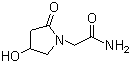
- C8H14N2O2
- Molecular Weight
- 170.21 g/mol
- Storage
- Store in a cool and dry place, away from light
- Poisonous
- NO
- Melting Point
- 112-116C
- Loss on Drying
- Not more than 0.5%
- HS Code
- 29242990
- Smell
- Odorless
- Shelf Life
- 2 years
- Color
- White to off-white
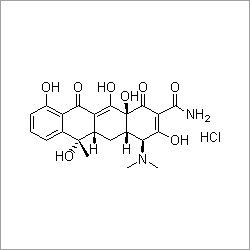
- Structural Formula
- C8H14N2O2
- Heavy Metal (%)
- Not more than 0.001%
- Solubility
- Freely soluble in water
- Medicine Name
- Levetiracetam API
- Chemical Name
- (S)--Ethyl-2-oxo-1-pyrrolidine acetamide
- CAS No
- 102767-28-2
- Type
- Other
- Grade
- Other
- Usage
- Pharmaceutical Intermediates, Used in the treatment of epilepsy
- Purity(%)
- 99% Min
- Appearance
- White to off-white crystalline powder
- Physical Form
- Powder
- pH (1% solution)
- 5.5 to 7.0
- Microbial Limit
- Complies with standard
- Assay
- 98.5% - 101.0%
- Specific Optical Rotation
- +105 to +112 (c=1 in water)
- Packing
- HDPE/Drum with double liners, 25kg net weight
- Residual Solvents
- Complies with ICH guidelines
- Related Substances
- NMT 0.5%
- Identification
- Complies with standard
- Shelf Life after Opening
- Use within 6 months
- Impurity Level
- < 0.1%
Levetiracetam Api Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
- Main Domestic Market
- All India
About Levetiracetam Api
Across the globe, our company is trusted as the best manufacturer and exporter of Levetiracetam Api which is used to treat epilepsy. Our product has a longer shelf life and is highly effective because of its accurate composition. All the quality oriented customers choose our company over others. They are highly pleased by the prices of our Levetiracetam Api. We give guarantee of accurate packaging of the product. Moreover, our company assures them about timely delivery of consignments.
Packaging Size: 1kg
Premium Quality and Compliance
Levetiracetam API is produced to surpass rigorous industry specifications and safety guidelines. Assured by compliance with ICH and local standards, it exhibits minimal impurities and heavy metal content. The API is tested for microbial safety, residual solvents, and provided in dust-free, secure packaging to ensure maximum integrity throughout the supply chain.
Versatile Usage in Epilepsy Treatment
Used predominantly as a pharmaceutical intermediate, Levetiracetam API forms the base for medications prescribed for epilepsy. Its high purity and stability make it an effective and reliable ingredient for anti-epileptic formulations, thereby supporting consistent therapeutic outcomes for patients.
Reliable Distribution and Handling
Packaged in 25kg HDPE drums with double liners for safety, Levetiracetam API is distributed, exported, and supplied globally by trusted manufacturers and traders based in India. Recommended storage in cool, dry, and light-protected environments safeguards its efficacy for up to 2 years, or 6 months after opening.
FAQs of Levetiracetam Api:
Q: How should Levetiracetam API be stored and handled to maintain its quality?
A: Levetiracetam API should be stored in a cool, dry place, away from direct light, and in its original HDPE drum with double liners. For best results, use the contents within 6 months after opening to ensure optimal potency and safety.Q: What are the primary benefits of using Levetiracetam API in pharmaceutical manufacturing?
A: The API offers high purity (minimum 99%), low impurity and heavy metal content (<0.1%, <0.001% respectively), and compliance with international safety standards, making it ideal for creating safe and effective medications for epilepsy.Q: When does Levetiracetam API expire, and what is its shelf life after opening?
A: Sealed Levetiracetam API has a shelf life of 2 years from the manufacturing date, provided it is stored appropriately. After opening, it is recommended to use the remaining product within 6 months.Q: Where is Levetiracetam API commonly supplied and who are the primary distributors?
A: This API is manufactured, supplied, and exported by licensed pharmaceutical companies and traders in India, and is further distributed worldwide for use in pharmaceutical formulations.Q: How is the quality of Levetiracetam API determined and ensured during production?
A: Quality is maintained through stringent testing for identity, assay (98.5%-101.0%), related substances (NMT 0.5%), heavy metals, microbial limit, and residual solvents, adhering closely to ICH and pharmacopeial guidelines.Q: What processes are involved in confirming the identification and purity of Levetiracetam API?
A: Identification is confirmed by standard analytical methods, including optical rotation and chemical tests. Purity is measured by assay, loss on drying, and impurity analysis, all aligned with pharmacopeial requirements.Q: What is the recommended medical application of Levetiracetam API?
A: Levetiracetam API is formulated primarily for medicines that manage epilepsy, offering reliable efficacy due to its consistent potency and chemical stability.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
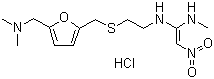
Oxiracetam .
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 62613825
Purity(%) : 99.0%
Usage : Nootropic agent, cognitive enhancer
Structural Formula : C6H10N2O3
Ranitidine hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 66357355
Purity(%) : 98.0% 102.0%
Usage : Used as a gastric acid reducer; Active Pharmaceutical Ingredient (API) in ulcer and GERD medications
Structural Formula : C13H22N4O3SHCl
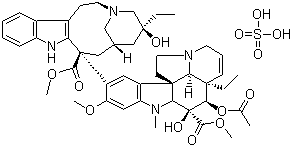
Vinblastine Sulfate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 143679
Purity(%) : >= 98%
Usage : Pharmaceutical raw material, anticancer agent
Structural Formula : (Refer to image for structure)
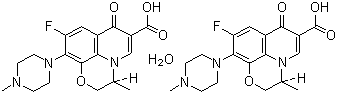
Levofloxacin hemihydrate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 138199710
Purity(%) : >= 98%
Usage : Pharmaceutical intermediate, active pharmaceutical ingredient (API), treatment of bacterial infections
Structural Formula : See image or chemical databases
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry