Oseltamivir Hydrochloride
Price 100 INR/ Kilograms
Oseltamivir Hydrochloride Specification
- Taste
- Bitter
- Particle Size
- 90% < 10 microns
- Loss on Drying
- Not more than 0.5%
- Molecular Formula
- C16H28N2O4 (as base)
- Color
- White to off-white
- Solubility
- Freely soluble in water, sparingly soluble in ethanol
- Boiling point
- Not applicable (decomposes before boiling)
- Heavy Metal (%)
- NMT 0.001%
- Melting Point
- Approx. 215-220C
- Ph Level
- 4.0 - 6.0 (1% solution)
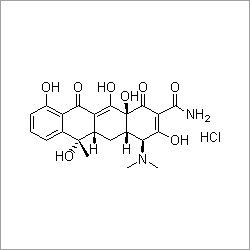
- Structural Formula
- See image
- Poisonous
- No (when used as indicated)
- Shelf Life
- 36 months
- Storage
- Store at 2C to 8C, protect from light and moisture
- Smell
- Odorless
- HS Code
- 29349990
- Molecular Weight
- 312.4 g/mol (as base), hydrochloride salt weight may differ
- Medicine Name
- Oseltamivir Hydrochloride
- Chemical Name
- (3R,4R,5S)-4-acetamido-5-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylic acid, ethyl ester, phosphate salt
- CAS No
- 204255-11-8
- Type
- Other
- Grade
- Other
- Usage
- Used as an antiviral agent for the treatment and prevention of influenza A and B.
- Purity(%)
- 98%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Residual Solvents
- Complies with ICH limits
- Endotoxin Level
- <0.25 EU/mg
- Specific Conductance
- <200 S/cm (1% solution)
- Optical Rotation
- +36 to +40 (c=1, H2O)
- Packing
- 1 kg, 5 kg, 10 kg HDPE drums or as required
- Related Substances
- Each impurity NMT 0.1%, total impurities NMT 0.5%
- Microbial Limits
- Total Aerobic Microbial Count <1000 CFU/g, Total Yeast & Mold <100 CFU/g
- Storage Conditions
- Keep tightly sealed in original container
- Identity Confirmation
- IR, HPLC, and Mass Spectrometry
- Impurity Profile
- Complies with pharmacopeial requirements
Oseltamivir Hydrochloride Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Oseltamivir Hydrochloride
Oseltamivir Hydrochloride: Versatile Antiviral for Broad Applications
Primarily used as an oral antiviral agent, Oseltamivir Hydrochloride is valued for its commercial significance in combating influenza A and B. Its usage type is pharmaceutical, and it is widely employed in clinical settings, hospitals, and healthcare facilities. The surface of application is internal, administered orally to prevent and treat flu infections. Its effectiveness across multiple commercial sectors makes it an essential component in the healthcare industry for managing seasonal outbreaks and ensuring public health.
Oseltamivir Hydrochloride: Sample & Packaging for Easy Exchange and Transportation
Samples of Oseltamivir Hydrochloride are readily available for evaluation, facilitating seamless exchange before large-scale procurement. The product is securely packaged in HDPE drums (1 kg, 5 kg, and 10 kg) for optimal preservation during transportation. Its versatility allows for smooth export market transactions, with the main domestic market being India. The packaging is customizable based on buyer needs, ensuring efficient handling and compliance with market demands both domestically and internationally.
FAQs of Oseltamivir Hydrochloride:
Q: How should Oseltamivir Hydrochloride be stored for maximum shelf life?
A: Oseltamivir Hydrochloride should be kept tightly sealed in its original container at 2C to 8C, protected from light and moisture, to ensure a shelf life of up to 36 months.Q: What are the recommended uses for Oseltamivir Hydrochloride?
A: This compound is used as an antiviral agent for the treatment and prevention of influenza A and B, especially in clinical, hospital, and pharmaceutical settings.Q: When is the best time to initiate treatment with Oseltamivir Hydrochloride for influenza?
A: Oseltamivir Hydrochloride is most effective when started within 48 hours of the onset of influenza symptoms, as recommended in medical guidelines.Q: Where can Oseltamivir Hydrochloride be exported from, and in what packaging sizes?
A: It is exported from India, available in packaging options of 1 kg, 5 kg, and 10 kg HDPE drums, with custom packaging upon request.Q: What is the process of confirming the identity and purity of Oseltamivir Hydrochloride?
A: Identity is confirmed through IR, HPLC, and Mass Spectrometry, and the product is tested to comply with pharmacopeial requirements for impurity profile, residual solvents, and microbial limits.Q: What are the commercial benefits of choosing this product from a reputable supplier?
A: Choosing Oseltamivir Hydrochloride from a trusted distributor ensures consistent quality, reliable supply, comprehensive documentation, and compliance with global pharmaceutical standards.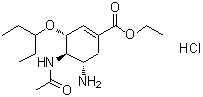

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Phloroglucinol
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Solubility : Freely soluble in water, ethanol, ether
Purity(%) : 99%
Heavy Metal (%) : 0.001%
Physical Form : Solid
Doxylamine succinate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Solubility : Soluble in water, ethanol, and chloroform
Purity(%) : 99%
Heavy Metal (%) : <0.001%
Physical Form : Solid
Calcitriol
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Solubility : Sparingly soluble in water, freely soluble in ethanol and chloroform
Purity(%) : 99%
Heavy Metal (%) : 0.001%
Physical Form : Solid
Minoxidil
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Solubility : Slightly soluble in water, soluble in propylene glycol, insoluble in chloroform
Purity(%) : 99% min
Heavy Metal (%) : 0.001%
Physical Form : Powder
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry