Pamidronate Disodium
Price 100 INR/ Kilograms
Pamidronate Disodium Specification
- Heavy Metal (%)
- <0.001%
- EINECS No
- 632-834-8
- Storage
- Store in a cool, dry place in tightly closed container
- Molecular Formula
- C3H9NNa2O7P2
- Loss on Drying
- <1.0%
- Shelf Life
- 2 years
- Molecular Weight
- 279.04 g/mol
- Melting Point
- >300C (decomposes)
- Ph Level
- 3.0 - 4.0 (1% solution)
- Poisonous
- Non-poisonous as raw material, for pharmaceutical use only
- Particle Size
- Micronized/powder, as specified
- Color
- White to off-white
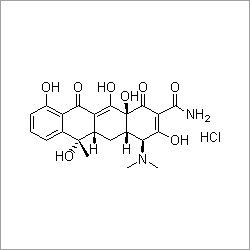
- Structural Formula
- C3H9NNa2O7P2
- HS Code
- 29319090
- Smell
- Odorless
- Solubility
- Soluble in water
- Medicine Name
- Pamidronate Disodium
- Chemical Name
- Pamidronate Disodium
- CAS No
- 94441-92-6
- Type
- Other
- Grade
- Other
- Usage
- Pharmaceutical raw material, used for the treatment of hypercalcemia and bone metastases
- Purity(%)
- >99%
- Appearance
- White to off-white powder
- Physical Form
- Powder
- Packing
- Double PE bag with aluminum foil outer drum, 1kg/5kg/10kg
- pH of 1% Solution
- 3.0 - 4.0
- Identification
- Conforms to standard
- Residual Solvent
- Meets ICH limits
- Assay (on dried basis)
- 99.0% - 101.0%
- Sulfate
- <0.05%
- Chloride
- <0.05%
- Endotoxin
- <5 EU/mg
- Total Impurities
- <0.5%
- Microbial Limit
- Total aerobic microbial count <1000 cfu/g
Pamidronate Disodium Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Pamidronate Disodium
Pamidronate Disodium: Usage and Superior Attributes
Pamidronate Disodium is utilized primarily as a pharmaceutical raw material for managing hypercalcemia and bone metastases. Its extra features include a splendiferous purity greater than 99%, low impurity profile, and micronized particle size for superior formulation. Suitable for plant-scale applications, it is offered in powder form that is highly soluble in water and conforms to rigorous microbial and heavy metal limits, ensuring the highest standards in medicinal manufacturing.
Sample, Payment Terms, and Certifications of Pamidronate Disodium
Samples of Pamidronate Disodium are available to ensure confident quality assessment prior to full outlay. Payment terms are flexible and tailored for seamless export market transactions. Delivered with requisite certifications, the product meets stringent ICH, EINECS, and microbial limit benchmarks. This guarantees compliance with international standards, making it a reliable choice for distributors, importers, and pharmaceutical manufacturers seeking assured transit and premium documentation.
FAQs of Pamidronate Disodium:
Q: How is Pamidronate Disodium commonly used in the pharmaceutical industry?
A: Pamidronate Disodium is primarily used as a raw material for the treatment of hypercalcemia and bone metastases. Its high purity and controlled particle size make it ideal for pharmaceutical formulations.Q: What are the key quality specifications of Pamidronate Disodium?
A: This product features an assay of 99.0% to 101.0%, total impurities less than 0.5%, and complies with ICH limits for residual solvents and microbial contamination, ensuring exceptional safety and efficacy.Q: When can samples of Pamidronate Disodium be requested?
A: Samples are available upon inquiry, allowing potential buyers to assess the products qualities and conformity before proceeding with a full-scale purchase or export transaction.Q: Where should Pamidronate Disodium be stored to maintain its stability?
A: Store the material in a cool, dry place within tightly closed containers to preserve its effectiveness and extend shelf life up to two years.Q: What are the benefits of choosing Pamidronate Disodium for medicinal manufacturing?
A: Its splendiferous purity, controlled impurity levels, and superior solubility offer reliable outcomes in pharmaceutical production, supporting stringent quality standards and patient safety.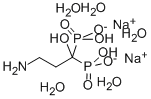

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
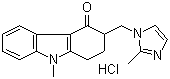
Ondansetron hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Smell : Other, Odorless
Solubility : Soluble in water and methanol; insoluble in ether
Structural Formula : C18H19N3OHCl
HS Code : 29420090
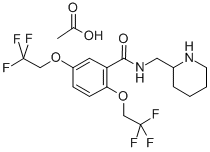
Flecainide Acetate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Smell : Other, Odorless
Solubility : Soluble in water and methanol
Structural Formula : C17H20F6N2O3C2H4O2 (displayed as per chemical structure diagram)
HS Code : 2924.29.98
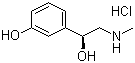
Phenylephrine Hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Smell : Other, Odorless
Solubility : Freely soluble in water, slightly soluble in alcohol
Structural Formula : C9H13NO2HCl
HS Code : 29334900
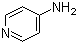
Dalfampridine
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Smell : Other, Odorless
Solubility : Soluble in water, slightly soluble in ethanol
Structural Formula : C5H6N2 (Pyrazine ring with amino substitution)
HS Code : 29333999
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry