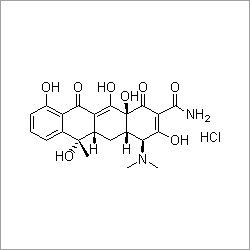
Ziprasidone hydrochloride
Price 100 INR/ Kilograms
Ziprasidone hydrochloride Specification
- Storage
- Store in tightly closed container, protected from light, at room temperature
- Molecular Weight
- 467.40 g/mol
- Boiling point
- Not applicable (decomposes)
- Heavy Metal (%)
- <0.001%
- Poisonous
- Yes, pharmaceutical use only under prescription
- Melting Point
- 242244C
- Molecular Formula
- C21H22ClN5OS HCl
- Loss on Drying
- <0.5%
- Particle Size
- Generally < 10 microns (customizable)
- Shelf Life
- 2 years from date of manufacture
- Color
- White to off-white
- Solubility
- Sparingly soluble in water; soluble in methanol and DMSO
- HS Code
- 29349900
- Ph Level
- Neutral to slightly acidic in solution
- Smell
- Odorless
- Structural Formula
- See image URL: https://cpimg.tistatic.com/4446601/s/4/Ziprasidone-hydrochloride.gif
- Taste
- Bitter
- Medicine Name
- Ziprasidone hydrochloride
- Chemical Name
- 5-[2-[4-(1,2-benzisothiazol-3-yl)-1-piperazinyl]ethyl]-6-chloro-1,3-dihydro-2H-indol-2-one hydrochloride
- CAS No
- 138982-67-9
- Type
- Other
- Grade
- Other
- Usage
- Used as an antipsychotic, primarily for the treatment of schizophrenia and bipolar disorder
- Purity(%)
- 99% (typical)
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Residual Solvents
- Meets ICH guidelines
- Moisture Content
- <0.5%
- Stability
- Stable under recommended storage conditions
- Impurities
- Single impurity <0.1%, Total impurities <0.5%
- Packaging Type
- Double polyethylene bags in fiber drums
- Therapeutic Category
- Atypical antipsychotic
- Identification
- Conforms to standard by IR/UV/HPLC
- Assay
- >98.5% (by HPLC)
Ziprasidone hydrochloride Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Ziprasidone hydrochloride
Application & Use of Ziprasidone Hydrochloride
Ziprasidone hydrochloride finds primary application in the therapeutic management of schizophrenia and bipolar disorder. Used exclusively under prescription, its role as an atypical antipsychotic enables effective psychiatric intervention. From pharmaceutical manufacturing to clinical supply, the product is reserved for elite medical use, ensuring safety and efficacy for patients who require unparalleled mental health care. Specialist distribution supports hospitals, pharmacies, and research institutions in delivering optimal therapeutic outcomes.
Sample Policy, Supply Ability & Export Markets
Samples of Ziprasidone hydrochloride are generally available upon request at a special sale price or proposal amount, ensuring accessibility for quality assessment. With substantial market value, the supply ability extends to bulk quantities for urgent or scheduled deliveries. The main export markets include North America, Europe, Asia, and the Middle East, underpinned by global demand and competitive clearance rates. Consistent delivery timelines and elite service standards make this an unrivaled choice for reliable supply.
FAQs of Ziprasidone hydrochloride:
Q: How should Ziprasidone hydrochloride be stored for optimal stability?
A: Ziprasidone hydrochloride should be stored in a tightly closed container, protected from light, at room temperature to maintain its stability throughout its two-year shelf life.Q: What is the main therapeutic use of Ziprasidone hydrochloride?
A: The primary usage of Ziprasidone hydrochloride is as an antipsychotic for the treatment of schizophrenia and bipolar disorder, prescribed strictly by licensed healthcare professionals.Q: When is a sample of Ziprasidone hydrochloride available for evaluation?
A: Samples are typically available at a special proposal amount or sale price upon request, allowing potential clients to assess the products quality and market value before making a bulk purchase.Q: Where can Ziprasidone hydrochloride be exported?
A: Ziprasidone hydrochloride is exported globally, with main markets in North America, Europe, Asia, and the Middle East, meeting stringent pharmaceutical requirements in each region.Q: What process assures the purity and identification of Ziprasidone hydrochloride?
A: Purity is rigorously tested by HPLC, and identification conforms to IR/UV/HPLC standards, ensuring that the product meets all critical pharmaceutical specifications and quality benchmarks.Q: How does the low impurity profile of Ziprasidone hydrochloride benefit users?
A: A single impurity of less than 0.1% and total impurities under 0.5% deliver a cleaner medicine, enhancing therapeutic outcomes and reducing risks of adverse reactions for patients.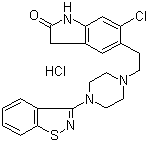

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
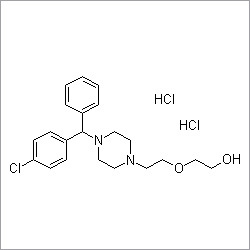
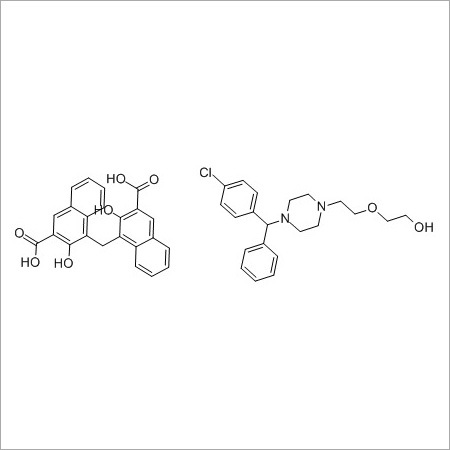
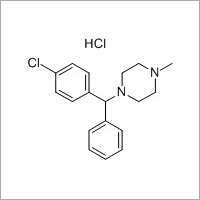
More Products in Pharmaceutical API Category
Ranitidine hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Ranitidine hydrochloride
Storage : Other, Store in a tightly closed container, protected from light and moisture at room temperature
Heavy Metal (%) : Not more than 10 ppm
Purity(%) : 98.0% 102.0%
Heptaminol hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Heptaminol hydrochloride
Storage : Other, Store in a cool, dry place, keep container tightly closed
Heavy Metal (%) : 0.001%
Purity(%) : 99.0%
Cyclobenzaprine Hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Cyclobenzaprine Hydrochloride
Storage : Other, Store in a cool, dry place, tightly sealed
Heavy Metal (%) : <20 ppm
Purity(%) : 99%
Neostigmine methyl sulfate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : Neostigmine methylsulfate
Storage : Other, Store below 30C in a tightly closed container
Heavy Metal (%) : 0.001%
Purity(%) : 99%
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry