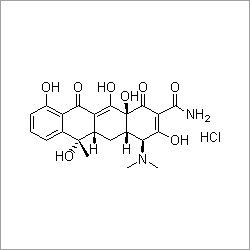
Diacerein
Diacerein Specification
- Melting Point
- 230C
- Shelf Life
- 3 years
- Ph Level
- Neutral
- Particle Size
- 10 microns
- HS Code
- 29349900
- Molecular Weight
- 368.30 g/mol
- Loss on Drying
- 0.5%
- Heavy Metal (%)
- 0.001%
- Storage
- Store in a cool, dry place, protected from light
- Boiling point
- Not applicable (decomposes)
- Structural Formula
- C19H12O8
- EINECS No
- 237-380-9
- Color
- Yellow
- Smell
- Odorless
- Poisonous
- Non-poisonous at therapeutic doses
- Solubility
- Slightly soluble in water, freely soluble in DMSO and methanol
- Molecular Formula
- C19H12O8
- Taste
- Bitter
- Medicine Name
- Diacerein
- Chemical Name
- Diacetylrhein
- CAS No
- 13739-02-1
- Type
- Other
- Grade
- Other
- Usage
- Used in the treatment of osteoarthritis
- Purity(%)
- 99%
- Appearance
- Yellow Crystalline Powder
- Physical Form
- Solid
- Stability
- Stable under recommended storage conditions
- Identification
- Complies with standard (IR/UV/HPLC)
- Pharmacopoeial Compliance
- USP/EP/IP
- Packing
- Double LDPE bags in HDPE drum
- Microbial Limits
- Below specified pharmacopeial limits
- Residual Solvents
- Complies with ICH guidelines
- Impurity Level
- 0.1%
- Use Category
- Chondroprotective agent
Diacerein Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Diacerein
Diacerein Famotidine is an aromatic organic compound with a molecular formula C19H12O8 and molar mass of 368.94 grams per mol. It is used for the treatment of arthritis that occurs when flexible tissue at the ends of bones wears down. It is a slow acting drug with properties like anti-inflammatory, anti-catabolic and pro-anabolic. Diacerein Famotidine works by decreasing expression of matrix metalloproteinase to maintain altered osteoblast activity. It is advised to take this medication after consulting and discussing your medical history with the doctor. It is not suitable for pregnant women. It is less effective and slow to cure pain.
Superior Quality & Pharmacopoeial Compliance
Diacerein supplied meets USP, EP, and IP standards, guaranteeing consistent potency and safety for pharmaceutical use. Rigorous impurity and heavy metal checks, along with compliance with ICH residual solvents guidelines, ensure maximum patient protection and performance reliability.
Ideal for Osteoarthritis Treatment
As a chondroprotective agent, Diacerein is extensively used in osteoarthritis management, providing symptomatic relief while supporting joint health. Its neutral pH, odorless nature, and controlled particle size allow for efficient formulation and patient compliance.
Secure and Robust Packaging
Double LDPE bags within an HDPE drum safeguard Diacerein from moisture, contamination, and light. These measures, paired with correct storage in cool, dry environments, help retain stability and efficacy for up to three years, supporting consistent medical supply chains.
FAQ's of Diacerein:
Q: How is Diacerein used in the treatment of osteoarthritis?
A: Diacerein is primarily administered as an oral medication to reduce inflammation and slow cartilage degradation in people with osteoarthritis. It offers both symptomatic relief and potential disease-modifying effects by protecting joint cartilage.Q: What makes Diacerein compliant with international pharmacopoeial standards?
A: Diacerein meets pharmacopoeial specifications set forth by USP, EP, and IP thanks to robust quality control measures, including strict limits on impurities (0.1%), heavy metals (0.001%), and comprehensive identification by IR, UV, and HPLC methods.Q: When should Diacerein be stored and what storage conditions are recommended?
A: It should be stored immediately upon receipt in a cool, dry place, protected from light. Following recommended storage conditions helps maintain its stability and efficacy for its entire three-year shelf life.Q: Where does the manufacturing and supply of Diacerein primarily occur?
A: Diacerein is manufactured, supplied, exported, and distributed widely across India by authorized manufacturers, suppliers, traders, and distributors, ensuring reliable product availability domestically and internationally.Q: What is the benefit of Diacerein's specialized packaging?
A: The product is packaged in double LDPE bags inside HDPE drums. This secure packaging ensures protection from moisture, contaminants, and light exposure, which maintains its physical and chemical stability during storage and transportation.Q: How does the low impurity and microbial levels in Diacerein benefit users?
A: With impurities at only 0.1% and microbial limits below pharmacopeial thresholds, Diacerein ensures safety, minimizes potential side effects, and guarantees high therapeutic quality for end users.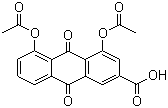
Price 100 INR/ Kilograms
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
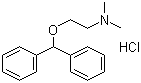
Diphenhydramine hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Purity(%) : 99%
Loss on Drying : 0.5%
Heavy Metal (%) : 0.001%
Storage : Other, Store in a cool, dry place in tightly closed container
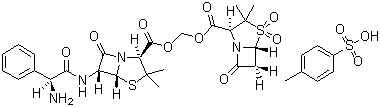
Sultamicillin tosilate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : >99% (HPLC)
Loss on Drying : 1.0%
Heavy Metal (%) : 0.001%
Storage : Other, Store in a cool, dry place away from direct sunlight
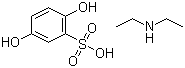
Etamsylate
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : 98%
Loss on Drying : 0.5%
Heavy Metal (%) : 0.001%
Storage : Other, Store in a cool, dry place away from light
Linezolid APi
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : 99%
Loss on Drying : 0.5%
Heavy Metal (%) : 0.002%
Storage : Other, Store in cool, dry, wellventilated area, tightly closed, protected from moisture
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |