Dicloxacillin sodium
Dicloxacillin sodium Specification
- Melting Point
- >220C (decomposes)
- Smell
- Odorless
- Molecular Formula
- C19H16Cl2N3NaO5S
- HS Code
- 29419090
- Shelf Life
- 2-3 years if properly stored
- Color
- White to off-white
- Loss on Drying
- 2.0%
- Solubility
- Freely soluble in water, sparingly soluble in alcohol
- Storage
- Store in a cool, dry place away from light and moisture
- Taste
- Bitter
- Poisonous
- No (when used as directed)
- Molecular Weight
- 510.31 g/mol
- Heavy Metal (%)
- 0.001%
- Particle Size
- Passes through 40 mesh
- Boiling point
- Not applicable (decomposes before boiling)
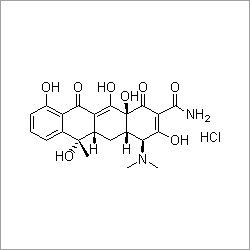
- Structural Formula
- See image in the given URL
- Ph Level
- 6.08.0 (10% aqueous solution)
- EINECS No
- 236-792-8
- Medicine Name
- Dicloxacillin Sodium
- Chemical Name
- (2S,5R,6R)-6-[(2,6-dichlorophenyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid sodium salt
- CAS No
- 13498-10-1
- Type
- Other
- Grade
- Other
- Usage
- Pharmaceutical intermediate, active pharmaceutical ingredient (API)
- Purity(%)
- Not less than 98.0%
- Appearance
- White or almost white powder
- Physical Form
- Solid
Dicloxacillin sodium Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Dicloxacillin sodium
As an API (active pharmaceutical ingredient), dicloxacillin sodium is the primary active ingredient in a medication. It is typically produced as a powder or crystalline solid, and is used in the production of dicloxacillin sodium capsules, tablets, and injections. Dicloxacillin sodium is an antibiotic drug that belongs to the class of penicillinase-resistant penicillins. It is used to treat a variety of bacterial infections, including skin and soft tissue infections, respiratory tract infections, and urinary tract infections. Dicloxacillin sodium API is typically produced through a multi-step synthesis process that involves the reaction of 6-amino penicillanic acid (6-APA) with a side chain that is specific to dicloxacillin. This reaction produces dicloxacillin sodium, which is then purified and isolated for use in pharmaceutical formulations.
High Purity and Quality Assurance
Dicloxacillin Sodium is manufactured according to international quality standards, ensuring chromatographic purity of at least 98.0% and compliance with residual solvent, microbial, and heavy metal regulations. It undergoes rigorous HPLC analysis and quality checks, making it a reliable choice for pharmaceutical manufacturers and researchers.
Suitable Packaging for Protection and Convenience
The products packaging in HDPE drums with double polyethylene liners secures it against moisture and contamination during storage and transit. Available sizes include 5kg, 10kg, and 25kg, catering to varying user requirements while maintaining product integrity.
Versatile Applications in Pharmaceuticals
Primarily used as an API and pharmaceutical intermediate, Dicloxacillin Sodium plays a critical role in the formulation of antibiotic medications. Its excellent solubility and high purity make it suitable for a wide array of pharmaceutical preparations, ensuring consistent results and efficacy.
FAQs of Dicloxacillin sodium:
Q: How is Dicloxacillin Sodium typically used in pharmaceutical applications?
A: Dicloxacillin Sodium is mainly employed as an active pharmaceutical ingredient (API) and as an intermediate in the production of antibiotics, valued for its high purity and consistent pharmacological profile.Q: What are the storage recommendations for Dicloxacillin Sodium?
A: It is recommended to store Dicloxacillin Sodium in a cool, dry place, away from direct light and moisture. Proper storage helps maintain its stability, quality, and shelf life of 23 years.Q: When should Dicloxacillin Sodium be tested for quality parameters?
A: Quality parameters such as purity, moisture content, endotoxin level, and microbial limits should be verified upon receipt and periodically during storage, especially if any physical changes are noticed or prior to pharmaceutical use.Q: Where is this Dicloxacillin Sodium commonly supplied from?
A: This product is often supplied by manufacturers, exporters, and distributors in India, serving global pharmaceutical markets through reputable trading channels.Q: What is the process for ensuring compliance with ICH Q3C residual solvent guidelines?
A: Manufacturers monitor and control the use of solvents during production, followed by analytical testing to confirm residual levels comply with ICH Q3C guidelines before packaging.Q: Which benefit does its particle size and solubility provide?
A: Passing through a 40 mesh and being freely soluble in water, Dicloxacillin Sodium ensures efficient processing in pharmaceutical formulations and rapid dissolution, contributing to better product performance.Q: Is Dicloxacillin Sodium safe to handle as a powder?
A: When used as directed and handled with appropriate safety precautions, Dicloxacillin Sodium is not considered poisonous or hazardous, though standard laboratory safety practices should always be followed.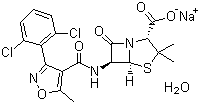
Price 100 INR/ Kilograms
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Terconazole .
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : ()cis1[4(2,4Dichlorophenoxy)phenyl]4,4dimethyl1,2,3triazolin5one
Melting Point : 128130C
Smell : Other, Odorless
Purity(%) : 99%
Camptothecin
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : (S)(+)Camptothecin
Melting Point : 267269C
Smell : Other, Odorless
Purity(%) : >98%
Isosorbide dinitrate
Price 100 INR / Kilograms
Minimum Order Quantity : 5 Kilograms
Chemical Name : Isosorbide dinitrate
Melting Point : 7075C
Smell : Other, Odorless
Purity(%) : 99%
Doramectin
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Chemical Name : (4R)4deoxy4(methylamino)avermectin B1
Melting Point : 173175C
Smell : Other, Odorless
Purity(%) : >98%
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |