Vinblastine Sulfate
Price 100 INR/ Kilograms
Vinblastine Sulfate Specification
- HS Code
- 29349900
- Storage
- Store at 2-8C in a tightly closed container, protected from light
- Boiling point
- Not applicable (decomposes before boiling)
- Color
- White to off-white
- Loss on Drying
- <2.0%
- Solubility
- Soluble in water and methanol; slightly soluble in ethanol
- Particle Size
- Micronized (as per manufacturer specification)
- EINECS No
- 205-606-0
- Shelf Life
- 2 years under recommended storage
- Molecular Weight
- 909.04 g/mol
- Taste
- Bitter
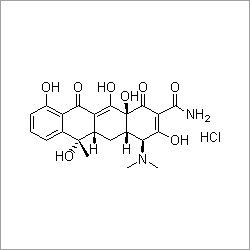
- Structural Formula
- (Refer to image for structure)
- Molecular Formula
- C46H58N4O9H2SO4
- Melting Point
- > 260C (decomposes)
- Poisonous
- Yes, toxic if ingested or improperly handled
- Heavy Metal (%)
- 0.001%
- Smell
- Odorless
- Medicine Name
- Vinblastine Sulfate
- Chemical Name
- Vinblastine sulfate
- CAS No
- 143-67-9
- Type
- Other
- Grade
- Other
- Usage
- Pharmaceutical raw material, anticancer agent
- Purity(%)
- >= 98%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Impurities
- Complies with pharmaceutical standards
- Therapeutic Category
- Antineoplastic (anti-cancer)
- Handling
- Use personal protective equipment, handle in a fume hood
- Standard
- USP/EP
- Stability
- Stable under recommended storage conditions
- Packing
- Custom as per requirement (typically vial or bottle)
- Application
- Used in treatment of various cancers including Hodgkin's lymphoma, testicular cancer, and breast cancer
- Identification
- Complies with reference standard by HPLC/IR spectroscopy
Vinblastine Sulfate Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Vinblastine Sulfate
Therapeutic Uses and Competitive Advantages
Vinblastine Sulfate is used as a frontline antineoplastic agent for treating various cancers, including Hodgkins lymphoma, testicular, and breast cancer. Its application spans hospital oncology wards and pharmaceutical manufacturing environments. The product stands out for its high purity, USP/EP compliance, and customizable packaging. Enhanced stability, precise impurity profiling, and strict adherence to international standards elevate it as a preferred choice among distributors, exporters, and healthcare institutions.
Certifications, Supply, and Domestic Availability
Vinblastine Sulfate is delivered with full certification, guaranteeing compliance with USP/EP and pharmaceutical impurity standards. Shipped worldwide by manufacturer, supplier, and trader networks, the product is always in supply due to a robust domestic mar and export infrastructure. Every batch is supported by a certificate of analysis, and orders are fulfilled promptly to ensure fast availability in all major domestic markets and international destinations.
FAQs of Vinblastine Sulfate:
Q: How should Vinblastine Sulfate be stored and handled safely?
A: Vinblastine Sulfate should be stored at 2-8C in a tightly closed container, protected from light. Use personal protective equipment and handle the substance in a fume hood to avoid exposure.Q: What therapeutic category does Vinblastine Sulfate belong to?
A: Vinblastine Sulfate falls under the antineoplastic (anti-cancer) therapeutic category and is primarily used for treating various types of cancer.Q: Where is Vinblastine Sulfate typically applied or administered?
A: It is applied in hospital oncology departments and pharmaceutical manufacturing settings as a raw material for anti-cancer medicine preparations.Q: What is the process for ensuring the purity and quality of Vinblastine Sulfate?
A: Purity (98%) and quality are assured by compliance with USP/EP standards and reference verification through HPLC/IR spectroscopy. Every batch undergoes rigorous impurity and stability testing.Q: When is Vinblastine Sulfate most beneficial in clinical settings?
A: It is most beneficial when included in chemotherapeutic regimens for cancers such as Hodgkins lymphoma, breast cancer, and testicular cancer, offering extensive therapeutic advantages.Q: What are the main benefits of choosing this product for pharmaceutical applications?
A: The main benefits include high purity, USP/EP standard compliance, custom packaging, superior stability, and a proven track record in effective cancer therapy.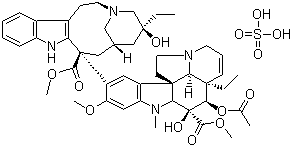

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
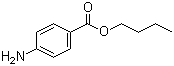
Butamben
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Medicine Name : Butamben
Molecular Weight : 193.24 g/mol
Shelf Life : 36 months
Structural Formula : C6H4(NH2)COOC4H9
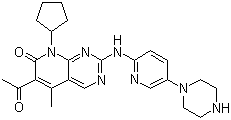
Paliperidone
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Medicine Name : Paliperidone
Molecular Weight : 426.48 g/mol
Shelf Life : 2 years from date of manufacture
Structural Formula : See product image/standard chemical structure.
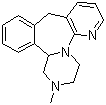
Mirtazapine
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Medicine Name : Mirtazapine
Molecular Weight : 265.36 g/mol
Shelf Life : 5 years under recommended storage conditions
Structural Formula : C17H19N3
Bromfenac
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Medicine Name : Bromfenac
Molecular Weight : 347.16 g/mol
Shelf Life : 2 years
Structural Formula : See image (bromfenac core structure)
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry