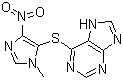
Ondansetron hydrochloride
Ondansetron hydrochloride Specification
- EINECS No
- 620-976-9
- Molecular Formula
- C18H19N3O HCl
- Shelf Life
- 3 years
- Color
- White to off-white
- Smell
- Odorless
- Poisonous
- Non-poisonous under prescribed usage
- Melting Point
- ~178-182C
- Storage
- Store in a cool, dry place, protected from light
- Heavy Metal (%)
- 0.001%
- Structural Formula
- C18H19N3OHCl
- Ph Level
- 4.0 - 6.0 (1% aqueous solution)
- Boiling point
- Not applicable (decomposes)
- Particle Size
- Micronized, as per specification
- Loss on Drying
- 0.5%
- Molecular Weight
- 329.83 g/mol
- Taste
- Slightly bitter
- Solubility
- Soluble in water and methanol; insoluble in ether
- HS Code
- 29420090
- Medicine Name
- Ondansetron Hydrochloride
- Chemical Name
- Ondansetron hydrochloride
- CAS No
- 99614-02-5
- Type
- Other
- Grade
- Other
- Usage
- Used for the prevention and treatment of nausea and vomiting caused by chemotherapy, radiation, or surgery
- Purity(%)
- >99%
- Appearance
- White to off-white crystalline powder
- Physical Form
- Solid
- Residue on Ignition
- 0.1%
- Microbial Limits
- Complies with pharmacopeial standards
- Identification
- IR/UV conforming to standard
- Regulatory Status
- DMF available upon request
- Packaging
- Available in HDPE drums of 1kg, 5kg, 10kg, or as required
- Chloride Content
- Conforms to standard
- Related Substances
- 0.1%
- USP/BP Compliance
- Meets USP/BP specifications
- Assay (on dried basis)
- >99.0% (HPLC)
Ondansetron hydrochloride Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
- Main Domestic Market
- All India
About Ondansetron hydrochloride
Application and Usage of Ondansetron Hydrochloride
Ondansetron Hydrochloride is typically administered orally or via injection, with application media being tablets, liquids, or injectable solutions. Its specific use lies in counteracting nausea and vomiting induced by chemotherapy, radiation therapy, or surgical procedures. More generally, it serves as an antiemetic for a wide range of patients. The product dissolves efficiently in water and methanol, ensuring ease of formulation and dependable results in various pharmaceutical preparations.
Certifications, Samples, and Payment Terms
Ondansetron Hydrochloride meets stringent USP/BP certifications and pharmacopeial microbial limits. For clients' convenience, free samples are available upon request, enabling thorough evaluation before bulk order. Cost-effective and flexible payment terms are offered. Express shipping and prompt freight delivery are assured worldwide. All documentation, including DMF, can be arranged, ensuring compliance and timely clearance for importers, distributors, and suppliers alike.
FAQ's of Ondansetron hydrochloride:
Q: How should Ondansetron Hydrochloride be administered for optimal results?
A: Ondansetron Hydrochloride can be taken orally as tablets or liquids, or administered as an injection, depending on medical advice and the patient's condition.Q: What are the primary benefits of using Ondansetron Hydrochloride?
A: It effectively prevents and treats nausea and vomiting caused by chemotherapy, radiation, or surgery, offering reliable symptom relief with a high purity profile.Q: When can I expect delivery of Ondansetron Hydrochloride after placing an order?
A: Express shipping and rapid freight options are available, ensuring prompt delivery of your order based on your location and shipping preference.Q: Where is Ondansetron Hydrochloride manufactured and supplied from?
A: This product is manufactured, exported, and supplied from India by established and certified manufacturers and distributors.Q: What certifications and quality standards does Ondansetron Hydrochloride comply with?
A: Ondansetron Hydrochloride fully complies with USP/BP standards and pharmacopeial microbial limits, and the Drug Master File (DMF) is available upon request.Q: Is it possible to obtain a sample of Ondansetron Hydrochloride for testing?
A: Yes, samples can be provided upon request, allowing clients to verify quality and suitability before committing to a bulk order.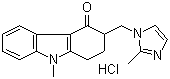
Price 100 INR/ Kilograms
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
- Main Domestic Market
- All India

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Azathioprine API
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : > 99%
Usage : Immunosuppressive agent used in organ transplantation and autoimmune disorders
Structural Formula : C9H7N7O2S (refer to official chemical databases for image)
Solubility : Slightly soluble in water, soluble in alkali
Iopamidol
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : >99%
Usage : Radiographic contrast medium for Xray imaging (CT, angiography, urography, etc.)
Structural Formula : See image
Solubility : Freely soluble in water; practically insoluble in ethanol, acetone, and methylene chloride
Tulathromycin A
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : >=98%
Usage : Veterinary antibiotic used mainly for the treatment of respiratory infections in cattle and swine.
Structural Formula : [See image or chemical reference for structure]
Solubility : Sparingly soluble in water, soluble in methanol
Fluphenazine dihydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : 99% (typical)
Usage : Pharmaceutical intermediate, antipsychotic agent
Structural Formula : See image
Solubility : Freely soluble in water, practically insoluble in ether
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |