Enoxaparin sodium
Enoxaparin sodium Specification
- Storage
- Store at 2C - 8C, protected from light
- Color
- White to off-white
- Solubility
- Freely soluble in water
- Melting Point
- Decomposes
- EINECS No
- 232-692-7
- Poisonous
- No (when used as directed)
- Ph Level
- 5.0 - 7.5 (1% solution)
- Heavy Metal (%)
- <0.001%
- Loss on Drying
- <10%
- Particle Size
- Fine powder
- Shelf Life
- 36 Months
- Structural Formula
- Complex polysaccharide structure (not representable as a simple formula)
- Smell
- Odorless
- HS Code
- 30012010
- Molecular Weight
- Approx. 4500-6500 Da
- Molecular Formula
- (C26H40N2O36S5)n
- Medicine Name
- Enoxaparin Sodium
- Chemical Name
- Enoxaparin Sodium
- CAS No
- 9005-49-6
- Type
- Other
- Grade
- Other
- Usage
- Anticoagulant, used for prevention and treatment of deep vein thrombosis and pulmonary embolism
- Purity(%)
- >99%
- Appearance
- White to off-white powder
- Physical Form
- Solid
- Registration
- DMF available
- Packing
- Sealed in sterile vials or bottles
- Standard
- USP/BP/EP
- Microbial Limits
- As per pharmacopeia standards
- Manufacture
- GMP certified facility
- Identification
- IR, UV, Chromatographic methods
- Storage Temperature
- 2-8C
- Assay
- >90 IU/mg anti-Xa activity
- pH of Solution
- 5.5 - 7.5
- Specific Optical Rotation
- +47 to +55 (c=1, H2O)
- Endotoxin Limit
- <0.05 IU/mg
- Sterility
- Sterile (for injectable forms)
Enoxaparin sodium Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Enoxaparin sodium
Enoxaparin Sodium Commercial Uses & Directions
Enoxaparin Sodium is acclaimed for its commercial use in hospitals and clinics, primarily aiding in the prevention and management of deep vein thrombosis (DVT) and pulmonary embolism. Suitable for administration under medical supervision, its dosage and method of injection must follow prescriber instructions, ensuring precision usage for optimal patient outcomes. Healthcare providers benefit from its fine powder form and reliable sterility, making it a preferred choice for regulated injectable medications.
Packaging, Certification & FOB Port Details
Packaging guarantees sterilityeach order is processed with vials or bottles sealed to preserve product integrity. Enoxaparin Sodium carries GMP certification, and DMF registration is available for seamless compliance. The FOB port is standardized for global transportation, accommodating distributor and importer needs. Inquire for asking price and packaging options to ensure tailored order processing. The product is shipped under controlled temperatures, with shelf life up to 36 months, facilitating convenient logistics and sustained potency.
FAQs of Enoxaparin sodium:
Q: How is the quality of Enoxaparin Sodium assured during manufacturing?
A: Enoxaparin Sodium is produced in a GMP certified facility, adhering to pharmacopeia standards with sterility and microbial limits strictly maintained throughout the manufacturing process. DMF registration is available for regulatory assurance.Q: What are the recommended usage directions for Enoxaparin Sodium?
A: Administer Enoxaparin Sodium solely under professional medical supervision, usually by injection. Dosage and schedule should align with healthcare guidelines for treating or preventing deep vein thrombosis and pulmonary embolism.Q: Where can Enoxaparin Sodium be ordered and shipped from?
A: Enoxaparin Sodium is available from global distributors, exporters, importers, and manufacturers. Shipping is facilitated via standardized FOB ports to ensure efficient international order processing.Q: What commercial benefits does Enoxaparin Sodium offer for healthcare facilities?
A: Enoxaparin Sodium stands out for its superlative purity, consistent anti-Xa activity, and ease of administration, making it reliable for both the prevention and treatment of thromboembolic disorders in regulated clinical environments.Q: How should Enoxaparin Sodium be stored to maintain its potency and safety?
A: Store Enoxaparin Sodium between 2C and 8C, protected from light, to preserve its chemical stability and sterility. When stored correctly, it offers a shelf life of up to 36 months.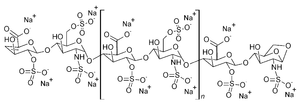
Price 100 INR/ Kilograms
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Aciclovir sodium
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 5536187
Usage : Antiviral agent, used for the treatment of herpes simplex and varicellazoster virus infections
Poisonous : Other, Nonpoisonous at therapeutic doses
Shelf Life : 2 years from date of manufacture when properly stored
Ambuphylline
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 359431
Usage : It is used as a bronchodilator in the treatment of asthma and COPD.
Poisonous : Other, Nonpoisonous at therapeutic doses
Shelf Life : 36 months
Hydroxyzine Hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 2192203
Usage : Antihistamine, Antiallergic, Anxiolytic
Poisonous : Other, No (when used appropriately in pharmaceutical doses)
Shelf Life : 23 Years
Cevimeline API
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 107233089
Usage : Treatment of dry mouth caused by Sjgrens syndrome
Poisonous : NO
Shelf Life : 2 years
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |