Aztreonam .
Price 100 INR/ Kilograms
Aztreonam . Specification
- HS Code
- 29419090
- Boiling point
- Not applicable (decomposes)
- Loss on Drying
- 1.0%
- EINECS No
- 278-826-5
- Solubility
- Soluble in water
- Melting Point
- >180C (decomposes)
- Storage
- Store below 25C, protected from light and moisture
- Shelf Life
- 2 years
- Poisonous
- NO
- Molecular Weight
- 435.44 g/mol
- Smell
- Odorless
- Particle Size
- Micronized
- Heavy Metal (%)
- 0.001%
- Color
- White to off-white
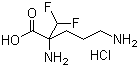
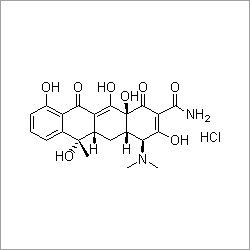
- Structural Formula
- C13H17N5O8S2
- Molecular Formula
- C13H17N5O8S2
- Medicine Name
- Aztreonam
- Chemical Name
- Aztreonam
- CAS No
- 78110-38-0
- Type
- Other
- Grade
- Other
- Usage
- Antibacterial
- Purity(%)
- 98% Min
- Appearance
- White to off-white powder
- Physical Form
- Powder
- Impurity Content
- 0.5%
- Packing
- Fiber drum with double polyethylene bags
- Application
- Treatment of infections caused by Gram-negative bacteria
- Stability
- Stable under recommended conditions
- Assay
- 98.0%~102.0%
- Endotoxin Level
- <1.0 EU/mg
- Pharmacopoeia Standard
- USP/EP/BP
- Residual Solvents
- Complies with ICH guidelines
- Optical Rotation
- +62 to +66
Aztreonam . Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Supply Ability
- 2000 Kilograms Per Day
- Delivery Time
- 1 Days
About Aztreonam .
Our product Aztreonam is highly trusted by manufacturers for making medicines to treat problems such as pneumonia and urinary tract infections. Numerous customers all over the world have been associated with our company. As per their feedback, no other suppliers and exporters offer high grade Aztreonam on these affordable prices. Therefore, they appreciate our efforts and admire our habit of keeping all the promises. We value their time and assure them about the quick as well as safe delivery of orders.
Pharmacopoeial Quality and Compliance
Aztreonam is produced in accordance with major international pharmacopoeia standards (USP, EP, BP), ensuring its suitability for regulated markets and therapeutic applications. Each batch is rigorously tested for assay, purity, and impurity content, maintaining high quality for end-users.
Applications and Efficacy
This medication is primarily used for the treatment of serious infections caused by Gram-negative bacteria. Its broad antibacterial spectrum makes it a trusted choice in clinical settings. Aztreonams reliable performance stems from its precise manufacturing and purity controls.
Safe Handling and Storage
To preserve Aztreonams stability and efficacy, store it in a cool environment below 25C, protected from light and moisture. Its non-toxic and odorless properties make handling safe. Proper storage extends shelf life up to two years.
FAQs of Aztreonam .:
Q: How should Aztreonam be stored to maintain its stability?
A: Aztreonam should be stored below 25C, shielded from light and moisture. Maintaining these conditions helps preserve its potency and extends its shelf life to up to two years.Q: What are the recommended applications of Aztreonam?
A: Aztreonam is primarily indicated for the treatment of infections caused by susceptible Gram-negative bacteria, including complicated intra-abdominal, urinary tract, and respiratory tract infections.Q: When should Aztreonam be used over other antibiotics?
A: Aztreonam is chosen when targeting Gram-negative bacterial infections, especially in cases where patients are allergic to penicillins or cephalosporins, as it offers a distinct safety profile.Q: Where is Aztreonam manufactured and supplied from?
A: Aztreonam is manufactured, exported, and supplied by facilities in India that comply with international pharmacopoeial standards and stringent quality guidelines.Q: What benefits does Aztreonam offer in clinical practice?
A: Aztreonams high purity, broad-spectrum activity, compliance with global standards, and low impurity content make it a dependable and effective antibacterial option in healthcare settings.Q: What is the process for ensuring Aztreonams quality?
A: Each batch of Aztreonam undergoes comprehensive testing for assay, impurity levels, residual solvents, heavy metals, and endotoxin levels to ensure compliance with USP/EP/BP standards before distribution.Q: How is Aztreonam packaged for distribution?
A: The product is securely packed in fiber drums with double polyethylene bags to protect it from moisture, light, and contamination during storage and transport.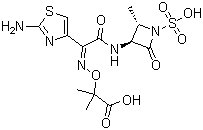

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical API Category
Eflornithine Hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 6640240
HS Code : 2924 29 95
Medicine Name : Eflornithine Hydrochloride
Melting Point : > 260C (decomposes)
Thiamine Hydrochloride
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 67038
HS Code : 29362500
Medicine Name : Thiamine Hydrochloride
Melting Point : 248250C (decomposes)
Bifonazole
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 60628968
HS Code : 29332990
Medicine Name : Bifonazole
Melting Point : 145147 C
Moxidectin
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
CAS No : 113507065
HS Code : 29329900
Medicine Name : Moxidectin
Melting Point : 141146C
 |
ANGLE BIO PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 Send Inquiry
Send Inquiry